Cholesterol Metabolism
Lipids = cholesterol and triglyceride – are insoluble in plasma and are transported in lipoproteins.
Functions = energy utilization, steroid hormone production, bile acid production, lipid deposition.
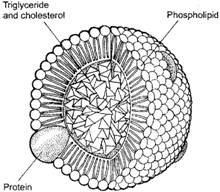
Major lipoproteins include
- Chylomicrons – large particles that carry dietary lipid
- Very low density lipoprotein – carry endogenous triglyceride and some cholesterol
- Intermediate density lipoprotein – carry cholesterol esters and triglycerides
- Low density lipoprotein – carry cholesterol esters
- High density lipoprotein – carry cholesterol esters
Exogenous pathway for lipid metabolism
- Dietary cholesterol and fatty acids are absorbed.
- Triglycerides are formed in the intestinal cell from free fatty acids and glycerol and cholesterol is esterified.
- Triglycerides and cholesterol combine to form chylomicrons.
- Chylomicrons enter the circulation and travel to peripheral sites.
- In peripheral tissues, free fatty acids are released from the chylomicrons to be used as energy, converted to triglyceride or stored in adipose.
- Remnants are used in the formation of HDL.
Endogenous pathway for lipid metabolism
- VLDL is formed in the liver from triglycerides and cholesterol esters.
- These can be hydrolyzed by lipoprotein lipase to form IDL or VLDL remnants.
- VLDL remnants are cleared from the circulation or incorporated into LDL.
- LDL particles contain a core of cholesterol esters and a smaller amount of triglyceride.
- LDL is internalized by hepatic and nonhepatic tissues.
- In the liver, LDL is converted into bile acids and secreted into the intestines.
- In non hepatic tissues, LDL is used in hormone production, cell membrane synthesis, or stored.
- LDL is also taken up by macrophages and other cells which can lead to excess accumulation and the formation of foam cells which are important in plaque formation.
HDL
HDL is a small particle composed of phospholipid and apolipoproteins and produced in hepatic and intestinal cells.
Benefits of HDL
Incidence of coronary heart disease events in a normal population is inversely related to the serum HDL-cholesterol concentration – low levels carry an increased coronary risk
HDL is thought to be anti-atherogenic and high HDL levels are cardioprotective.
This effect may be mediated by reverse cholesterol transport, a process whereby excess cholesterol in cells and in atherosclerotic plaques is removed and transported back to the liver.
Risk for myocardial infarction increases by about 25 percent for every 5 mg/dL decrement in serum HDL-cholesterol below median values for men and women.
Low HDL-cholesterol is a component of the metabolic syndrome that is characterized by obesity, insulin resistance, dyslipidemia, and hypertension
Patients considered high risk for cardiovascular disease based on HDL levels include:
- Patients with HDL less than 40 mg/dL
- Patients with the metabolic syndrome – gender adjusted HDL-cholesterol levels of less than 40 mg/dL in men and 50 mg/dL in women.
Exercise, weight loss (in overweight subjects), smoking cessation, and changes in diet (specifically substitution of monounsaturated for saturated fatty acids) all can raise HDL-cholesterol.
Medical treatment for low HDL includes niacin and fibrates.
SYNTHESIS OF CHOLESTEROL
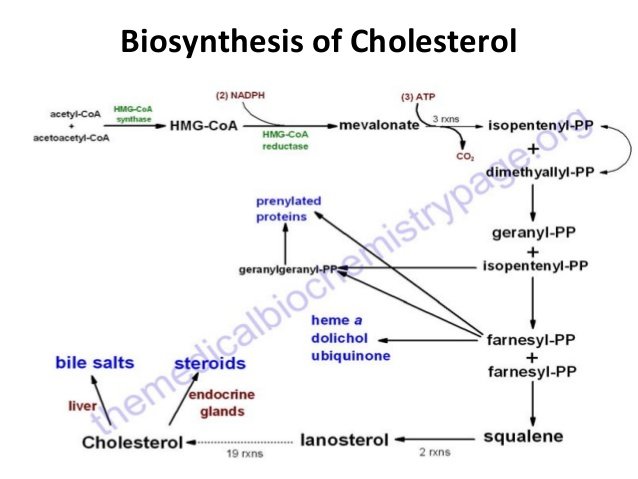
Cholesterol synthesis occurs in the cytoplasm and microsomes from the two-carbon acetate group of acetyl-CoA. 50% of the daily cholesterol production is converted to bile acids and salts and secreted in bile. Most of this re-absorbed and re-used by way of entero-hepatic circulation. About 0.5 – 1.0 g/day is used for the biosynthesis of steroid hormones (about 40 mg/day). A small part of cholesterol (about 50 mg/day) in adult is excreted also from the surface of the skin.
The process of cholesterol biosynthesis has five major steps:
Three molecules of acetyl-CoA are condensed and converted to 3-hydroxy-3-methyl-glutaryl-CoA (HMG-CoA).
HMG-CoA reductase catalyzed the conversion of HMG-CoA to mevalonate is the rate –limiting step in cholesterol biosynthesis in which cholesterol act as an allosteric inhibitor of gene expression of the enzyme.
Mevalonate is converted in several enzymatic steps to the isoprene based molecule, isopentenyl pyrophosphate (IPP) and is the key five-carbon isoprenoid intermediate in cholesterol biosynthesis, with the concomitant loss of carbon dioxide. IPP is also precursors in the synthesis of coenzyme Q (ubiquinone) and dolichol , which function in the synthesis of carbohydrate side chains in glycoproteins and another important biomolecules such as vitamin E , K and carotinoids.
Squalene, a 30-carbon molecule is formed by several condensation reactions involving isopentenyl ptrophosphate
Conversion of Squalene to cholesterol (a 27- carbon molecule) requires several reactions , some of them with unknown mechanism till now and involved the reducing equivalent NADPH and molecular oxygen ( mixed – oxygenase system).
The acetyl-CoA utilized for cholesterol biosynthesis is derived from an oxidation reaction (e.g., fatty acids or pyruvate) in the mitochondria and is transported to the cytoplasm by the same process as that described for fatty acid synthesis. Acetyl-CoA can also be derived from cytoplasmic oxidation of ethanol by acetyl-CoA synthetase. All the reduction reactions of cholesterol biosynthesis use NADPH as a cofactor.
CHOLESTEROL METABOLISM
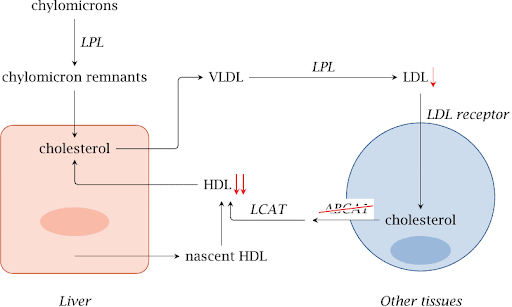
Cholesterol is transported in the plasma predominantly as cholesteryl esters associated with lipoproteins. Dietary cholesterol is transported from the small intestine to the liver within chylomicrons. Cholesterol synthesized by the liver, as well as any dietary cholesterol in the liver that exceeds hepatic needs, is transported in the serum within LDLs. The liver synthesizes VLDLs and these are converted to LDLs through the action of endothelial cell-associated lipoprotein lipase. Cholesterol found in plasma membranes can be extracted by HDLs and esterified by the HDL-associated enzyme LCAT. The cholesterol acquired from peripheral tissues by HDLs can then be transferred to VLDLs and LDLs via the action of cholesteryl ester transfer protein (apo-D) which is associated with HDLs. Reverse cholesterol transport allows peripheral cholesterol to be returned to the liver in LDLs. Ultimately, cholesterol is excreted in the bile as free cholesterol or as bile salts following conversion to bile acids in the liver.






where is Ldl Metabolism